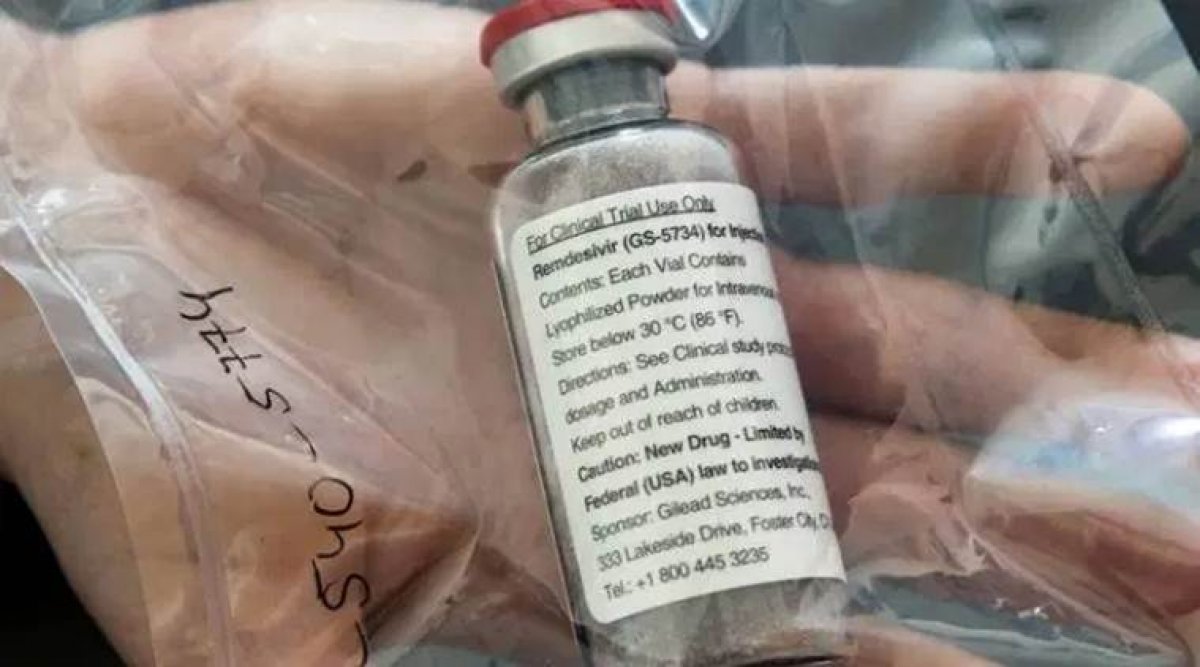
India will soon market and distribute the domestically produced Remdesivir drug for “restricted emergency use” for the treatment of coronavirus patients, according to statements from Indian companies.
Indian drugmakers CIPLA and Hetero Pharma launched the generic Remdesivir drugs production late Sunday, a month after signing the deal with the US-based patent-holder Gilead Sciences.
THE DRUG WILL NOT BE AVAILABLE ON COUNTERS
With total confirmed cases of coronavirus soaring at 425,282, the companies will be making the generic drugs available in the capital Delhi and the financial capital Mumbai first, the two worst-hit cities in the country, via hospital channels.
“At present, use of these therapies is based on limited available evidence. As the situation evolves, and when more data become available, the evidence will be accordingly incorporated, and recommendation upgraded,” according to the protocol.

CIPLA has launched the drug under the brand name “Cipremi”, while Hetero under the brand name “Covifor”.
Both companies have already received approval from the country’s Drug Controller General, as part of their agreement with Gilead Sciences.













